摘要:2025年4月2日,Denali Therapeutics 宣布已经启动递交 Tividenofusp Alfa(DNL310)的加速批准上市申请,用于治疗亨特氏综合征(MPS II)。
text:

After some high-profile crashes, the one-time biotech darling is inching toward success with its Hunter syndrome treatment, which today began a rolling BLA for accelerated approval.
Denali Therapeutics has its wheels on the fast track. The company initiated a biologics license application Wednesday for accelerated approval for Hunter syndrome treatment tividenofusp alfa (DNL310). It’s been a long time coming for Denali, which launched in 2015 with what was then the largest first-round venture financing in biotech history, a whopping $215 million. But a decade later, Denali doesn’t yet have an approved product, and its stock has dipped from its 2020 peak of more than $85 to just over $12 today.
“When we came out of stealth in 2015, we raised a significant amount of money,” Carole Ho, Denali’s chief medical officer and head of development, told BioSpace. “And there was a question: What were we doing differently?” The brand-new company trumpeted a lofty goal of deploying new approaches to tackle neurodegenerative diseases—by targeting newly discovered genes tied to degenerative brain diseases.
A year after launching, Denali scored another $130 million in a series B round led by Genentech veterans and maintained all of its founding investors. The company signed a bevy of research agreements, including deals with Blaze Bioscience and the Fred Hutchinson Cancer Research Center, to work on one of neurology’s toughest problems: getting drugs across the blood-brain barrier (BBB).
Now, Denali is eyeing the culmination of these approaches with DNL310, an enzyme-replacement therapy that rides a specialized transport vehicle across the blood-brain barrier. The therapy secured FDA Breakthrough Therapy Designation in January, adding to a list of honors already held by the drug, including Fast Track Designation, Orphan Drug Designation and Rare Pediatric Disease Designation.
“All of this suggests the agency’s enthusiasm for the molecule,” Ho said.
Stifel analysts responded to the BLA submission Wednesday morning, saying, “We’re potentially nearing the end of an up-and-down regulatory path for [DNL310], where DNLI was finally able to align with FDA’s CDER on leveraging CSF heparan-sulfate (HS) as a surrogate biomarker for the basis of accelerated approval.”
Stifel also noted the turbulent times in which this application has been submitted.
“Notably, this announcement comes amidst significant turnover at FDA, where several senior directors across both CBER/CDER have departed,” the analysts wrote.
……(节选至此,加入听友群,可获得完整文稿,并为你解答该药物的设计原理)
原文链接:www.biospace.com
药物原理:
DNL310 运用 ETV 递送透过血脑屏障
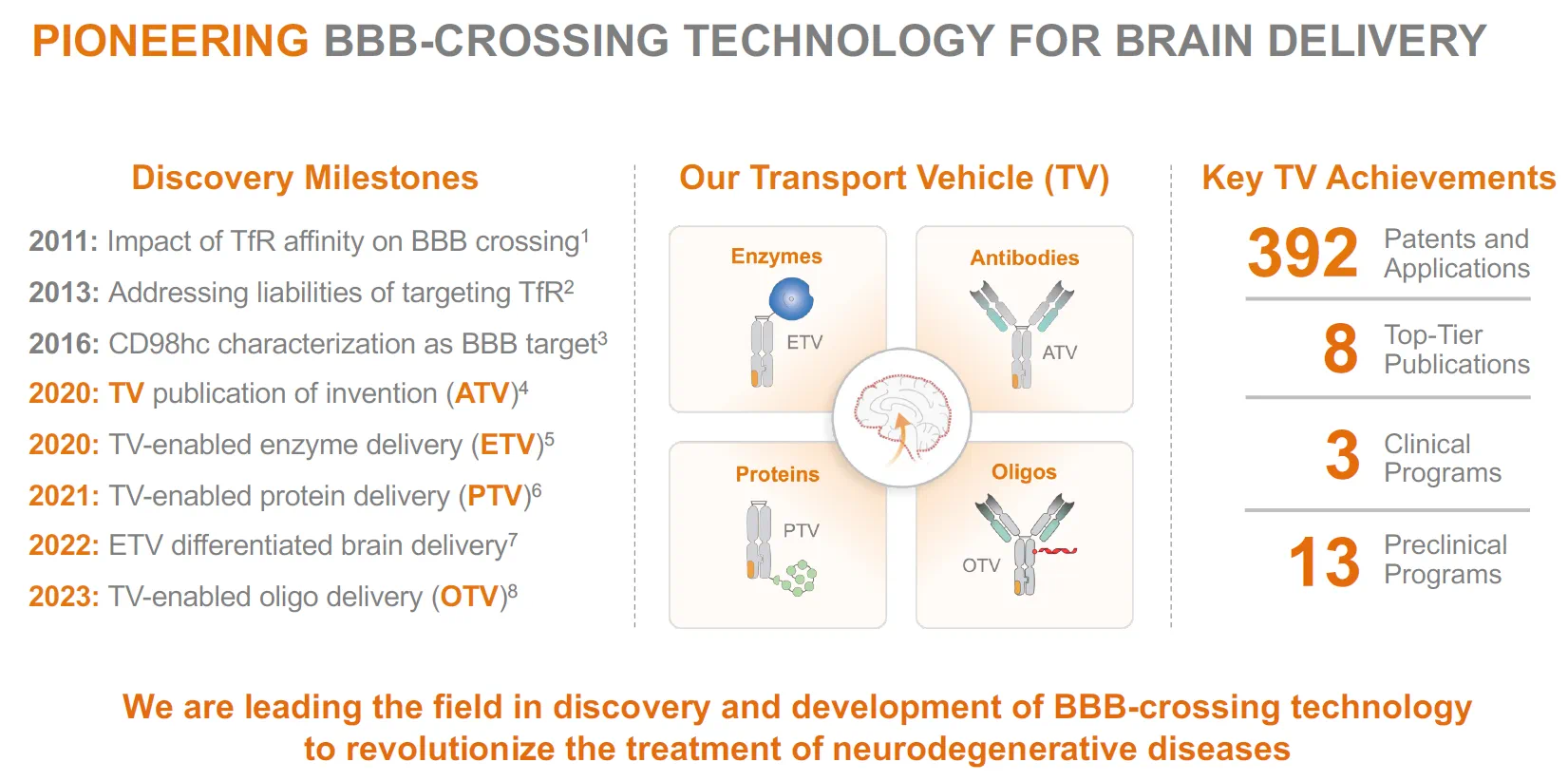
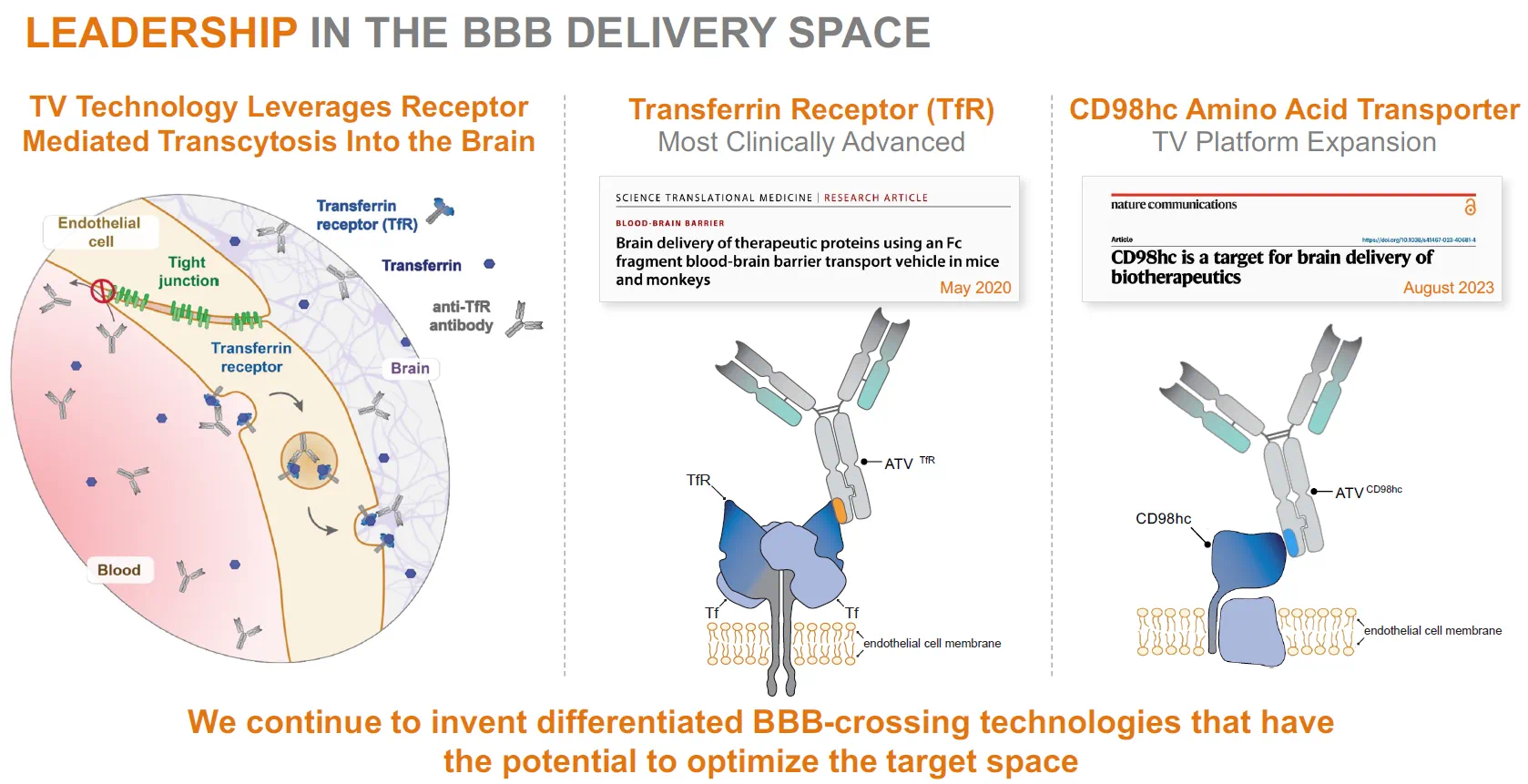
词汇:thorny adj. 棘手的;high-profile adj. 备受瞩目的;inch v. 缓慢移动;BLA, biologics license application abbr. 生物制剂许可申请;whop adj. 巨大的;dip v. 下降;come out 首次亮相;stealth n. 隐身;trumpet v. 鼓吹;lofty adj. 高远的;launch v. 启动;bevy n. 一堆东西;culmination n. 最终结果;pediatric adj. 小儿科的;heparan-sulfate n. 乙酰肝素;surrogate adj. 替代的
欢迎大家在评论区与我互动,给我写邮件1978500617@qq.com,或加入听友群 Daily Life Science group 1

欢迎大家关注我的小宇宙读书播客《启真书汇》,将陆续更新政治历史、经济金融、管理学、哲学、心理学和社会学等书籍的深度解读,月更。
欢迎大家关注我的微信公众号《思无涯-Gilly》,代谢与免疫前沿研究文献深度解读,月更。

本期制作:Gilly
热爱可抵岁月漫长~



 44
44 1
1
